Metronidazole: To BID or not to BID
Should we be dosing metronidazole three times a day or twice daily for common infections? We run through the data to help answer this question!

Yes, this is a Hamlet reference for all the Shakespeare fans out there.
We have a quick clinical dosing post for you today! Our focus is on the nitroimidazole, metronidazole, or better known as Flagyl. Metronidazole is a prodrug that is activated after bacterial uptake and reduction. To elicit a bactericidal effect, metronidazole diffuses into the organism, interacts with the bacterial DNA, causes a loss of helical DNA structure and strand breakage, and ultimately inhibits protein synthesis and causes cell death (Lexicomp 2023). Metronidazole is effective against anaerobic bacteria. "Anaerobic bacteria are considered normal human flora in the oral and intestinal spaces but are frequently implicated as causative pathogens when infection occurs at these sites." (Soule 2018)
In adults, time to peak for an oral tablet is around 1 to 2 hours. The half-life (how long it takes for the body to eliminate 50% of the drug) of metronidazole is roughly 8 hours. Therefore, the standard dosing regimen at some institutions is 500 mg every 8 hours (TID). Taking a tablet every 8 hours can be burdensome on a patient and also on nursing staff in the inpatient setting.
There are some indications for 500 mg every 12 hour (BID) dosing. I have listed these below:
- Bacterial vaginosis
- Crohn disease (adjunctive agent for treatment of simple perianal fistualas)
- Giardiasis
- Helicobacter pylori treatment
- Acute pouchitis
- Sexually transmitted infections
This means that the 12 other indications for metronidazole (listed on Lexicomp) recommend TID or QID (four times a day) dosing.
Some questions to ponder:
- Why are some regimens BID and some more frequent?
- What if there were pharmacokinetic and clinical studies that evaluated a TID regimen and a BID regimen?
Well, if you did not already guess it, there are studies! Let's look into some of the data behind a twice daily regimen for metronidazole and if it is an appropriate frequency to use in clinical practice.
Fun fact: My institution recently changed from every 8 hour dosing to every 12 hours*, and I thought it would be a neat update to share!
*exceptions exist; see below.

Pharmacokinetic Data
The first thing to consider when looking into dosing frequency is the pharmacokinetic properties of the drug. Pharmacokinetics refers to how the body absorbs, distributes, metabolizes, and eliminates the drug. Based on these parameters, we can assess time to peak concentration, how long a therapeutic concentration of drug is present in the body from one dose, and how fast the drug is eliminated.
In 1989, Earl et al. conducted a study looking at an every 12 hour dosage schedule for oral and intravenous (IV) metronidazole. They examined paired blood samples from 48 patients who received metronidazole every 12 hours. Twenty-five patients were given 400 mg metronidazole orally BID and 23 received 500 mg metronidazole IV BID.
Assay of the samples demonstrated trough serum concentrations of metronidazole of 3–11 mg/l (mean 5.5 mg/l) and 2–15 mg/l (mean 6.7 mg/l) with the oral and intravenous routes, respectively. The corresponding peak serum concentrations were 10–26 mg/l (mean 17.4 mg/l) and 13–28 mg/l (mean 23.6 mg/l). The trough concentrations are well in excess of the MICs for the majority of obligate anaerobes.
Below is a graphic representation of the serum peak and trough concentrations using data from Earl et al. and pharmacokinetic calculations to extrapolate concentrations from this data for a TID regimen using 500 mg IV dosing.
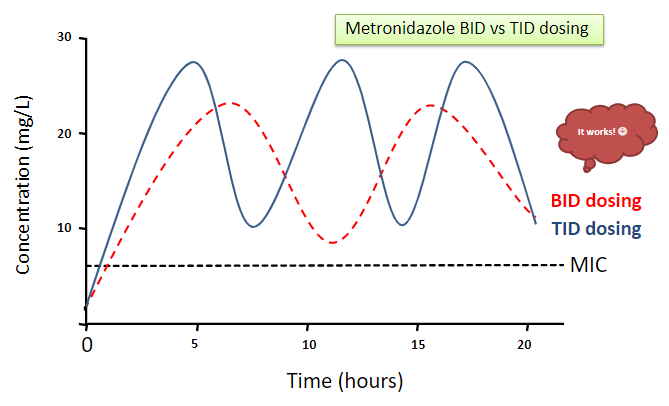
If BID dosing regimens were found to provide sufficient concentrations above the MIC for anaerobes in this study, then TID (more frequent) dosing would definitely provide high enough concentrations as well. This is represented by the figure above. The only concern now is what are the consequences (if any) of administering more drug than required to kill bacteria?
Clinical Data
The second thing to consider when looking into dosing frequency is the clinical data and how efficacious the regimen is at eradicating the infection.
There are two recent retrospective studies that looked at BID vs TID metronidazole dosing. The first study in 2018 evaluated metronidazole for the treatment of anaerobic or mixed anaerobic infections. Dose strength was not specified. Of the 200 patients included, no significant differences in clinical cure were seen between the groups (85% for both groups, p=1.00). The mean duration of therapy was 5.9 days in the BID group and 5.8 days in the TID group. Hospital length of stay was 8.1 days vs 6.7 days in the BID and TID groups, respectively (p > 0.05).
Most patients were being treated for abdominal infections (79% and 89%, respectively). The other infections treated were skin and soft tissue, pneumonia, and "other."
Overall, the authors of this study concluded that a BID regimen was an effective option to treat abdominal infections.
Another retrospective study published in 2022 looked at BID vs TID metronidazole dosing in Bacteroides spp. bloodstream infections. The primary endpoint was clinical failure (defined as a composite of all-cause 30-day mortality, escalation of antimicrobial therapy, 30-day readmission or recurrence due to an anaerobic infection, positive repeat blood cultures for Bacteroides spp., or failure to resolve leukocytosis or fever).
There were 208 patients included: 68 in the BID group and 140 in the TID group. The dose given in the study was 500 mg per dose. Overall, there was no significant difference between rates of clinical failure between the BID and TID groups. The authors conducted a multivariate analysis and found that neither the use of TID dosing, time to treatment initiation, days of initial non-metronidazole anaerobic therapy, pre-infection length of stay, admission prior to 2016, or initial oral metronidazole use were significantly associated with clinical failure.
From this study, the authors concluded that, "in the absence of a clear benefit, metronidazole 500 mg BID is a reasonable dosing strategy in lieu 500 mg TID for infections due to Bacteroides spp."
Final Thoughts and Clinical Application
The PK and clinical data together suggest that BID metronidazole dosing regimens are equally effective for several indications. The caveat to the BID dosing regimen is the following:
- C. difficile infections
- Central nervous system (CNS) infections
- Parasitic/amoebic infections
This is because these infections are more difficult to treat and require higher concentrations of metronidazole.
Metronidazole is associated with adverse effects due to elevated concentrations. Reducing dosing frequencies and total drug exposure will help limit the development of certain toxicities (specifically CNS related toxicities). In addition, switching to BID dosing will help ease medication burden on patients and nursing staff.
So, whether or not you currently practice BID dosing for metronidazole, now you know it is safe and effective to prescribe twice daily regimens instead of three times a day! AND you have the data to back it up if you are asked to explain your rationale.
Happy dosing!


