New Oral GLP-1 Orforglipron Receives FDA Approval
FDA-approved orforglipron is the first oral non-peptide GLP-1, offering weight loss and glycemic control without injections.

Just last month, the FDA approved orforglipron (Foundayo) – the first non-peptide glucagon-like peptide 1 (GLP-1) receptor agonist for glycemic control and weight loss.1
Introduction (and a little medicinal chemistry!)
GLP-1 receptor agonists are generally very large molecules. Semaglutide (Ozempic, Wegovy), for example, is a sequence of 31 amino acids with a molecular weight of 4,114 g/mol.2 Orforglipron has a molecular weight of 883 g/mol, a much smaller molecule than its predecessors. While impressive on its own, the comparatively small size of this molecule confers a number of benefits that previous GLP-1 therapies lack.
While GLP-1s have taken the world of type 2 diabetes (T2D) and weight management by storm, some patients may be reluctant to start injectable therapy. Orforglipron demonstrates similar efficacy to others in its class, and being available orally may give it an edge over injectable weight loss therapies like Wegovy or Mounjaro.3
“Glucagon-like peptide-1 receptor agonists have emerged as a cornerstone in the management of T2D and obesity, providing glucose-dependent insulin secretion, glucagon suppression, delayed gastric emptying, appetite reduction, and cardiometabolic benefits,” according to the 2026 article reviewing this new medication.3 “Oral formulations are highly desirable to improve patient acceptance and broaden therapeutic reach.”
Oral GLP-1s
Orforglipron is not the first oral GLP-1. That title goes to Rybelsus, an oral formulation of semaglutide. However, since semaglutide is a peptide, it is subject to cleavage and metabolism by the enzyme dipeptidyl peptidase-4.3 Orforglipron, being much smaller, is not a substrate for this enzyme, allowing it to bypass early metabolism other GLP-1s must overcome.3 Its size also offers protection from the acidic environment of the stomach, another obstacle to oral availability for other GLP-1s.3
“The compound contains strategically positioned polar functional groups that balance aqueous solubility with membrane permeability, a key requirement for efficient oral absorption,” according to the 2026 review article.3 “These polar elements participate in hydrogen bonding and electrostatic interactions within the receptor binding pocket, while surrounding lipophilic regions promote favorable pharmacokinetics and sustained GLP-1 receptor engagement.”
Basically, the drug developers ensured that part of the molecule is attracted to water, which helps the body absorb the medicine through the gastrointestinal tract. These same water-loving segments of the molecule also help keep it in the GLP-1 receptor for a longer period of time.
So What's the Benefit of Orforglipron?
Besides these differences, orforglipron has similar effects in the body as other GLP-1s. The ACHIEVE-1 trial focused on adults with diabetes4 and demonstrated a roughly 1.5% reduction in hemoglobin A1c (HbA1c) with no significant incidence of hypoglycemia, as well as a reduction in appetite secondary to interactions with the central nervous system.3
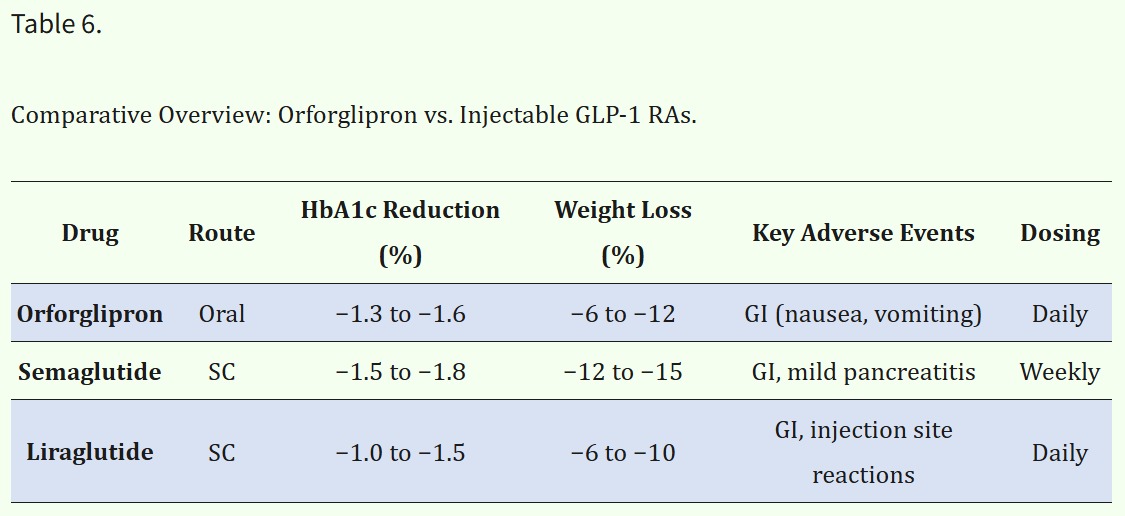
Additionally, the ATTAIN studies – which focused on adults with obesity4 – showed promising results for weight management in patients with diabetes and without: 12-15% reduction in non-diabetic patients, and 10-12% reduction in diabetic patients.3
So far, the biggest adverse effects are transient, dose-related gastrointestinal issues.3 Discontinuation due to adverse effects was rare.3 However, there is no data on potential long-term complications due to study durations of 26-72 weeks.3
The approval of an oral, non-peptide GLP-1 is a pioneering step in diabetes and weight management. Orforglipron looks promising, given the data available thus far. Time will tell if it holds up against its injectable predecessors.
References
1 Novel Drug Approvals for 2026. Food and Drug Administration. 2026.
2 Semaglutide. PubChem. National Center for Biotechnology Information. 2026.
4 What to know about orforglipron: An investigational oral GLP-1. Eli Lilly and Company. Lilly USA, LLC. 2026 Feb.
*Information presented on RxTeach does not represent the opinion of any specific company, organization, or team other than the authors themselves. No patient-provider relationship is created.

