The Return of Ranitidine
Ranitidine (Zantac) returns after NDMA-related recall, with reformulation aimed at improving safety while maintaining GERD treatment efficacy.
From 2004 to 2020, ranitidine graced over-the-counter shelves as the product Zantac, used to treat heartburn and gastroesophageal reflux disease (GERD).1 It was taken off the market for five years due to concerns with carcinogens, but as of November 2025, Zantac is back.
The rise
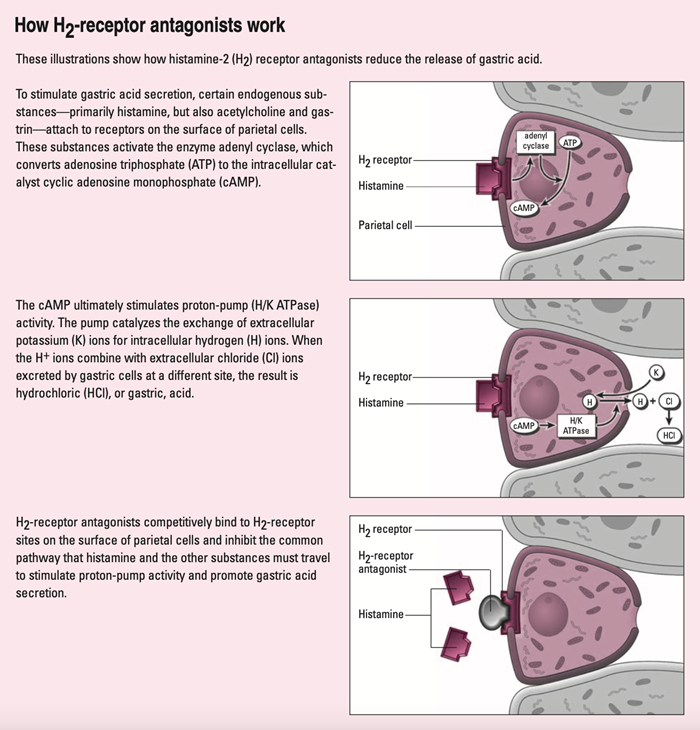
Ranitidine is a histamine type 2 receptor antagonist, reversibly blocking the production of hydrochloric acid in the stomach to provide relief from heartburn and GERD, as well as duodenal ulcers and other issues associated with gastric hyperacidity.2 Ranitidine works the same way as both cimetidine (Tagamet) and the much more popular famotidine (Pepcid). See Figure 1 below for a visual representation of the mechanism of action of these drugs.
Ranitidine was one of the most popular drugs of its time, first approved in 1983 for prescription use and in 2004 for over-the-counter (OTC) use.1 After just five years on the market, it became the world’s best-selling drug.3
The fall
But in September 2019, things unravelled for ranitidine. Impurities reported in ranitidine products caused pharmaceutical companies to stop distribution and retail pharmacy chains to remove them from shelves.3 The next month, the FDA reported “unacceptable” levels of N-nitrosodimethylamine (NDMA), a cancer-causing chemical.3
NDMA is primarily an industrial byproduct, once used in the production of rocket fuel before its toxic effects were realized.4 Humans are generally exposed to NDMA via contaminated food or drinking water, and severe overexposure can lead to liver dysfunction.4 It is officially classified as a “probable human carcinogen” and has been confirmed to induce tumors in animals in lab settings.4
After more testing found that NDMA accumulated in ranitidine with age and heat, the FDA finally removed ranitidine from the market in April 2020.5 The Zantac brand stuck around in the form of Zantac 360, which contained the active drug famotidine, like its Pepcid counterpart.6
The rebirth
More than five years later, in November 2025, the FDA announced that ranitidine would be back on the market. According to the FDA, the product had been “reformulated” but “maintains the same therapeutic effectiveness” as older products.7
The FDA shared the following advice for proper handling of the medication:
Keep ranitidine tablets in the original container (bottle) and protect from moisture.
After the first opening of the bottle, discard unused tablets after 3 months (90 days), or by the expiration date on the bottle, whichever is sooner.
If more than one bottle is dispensed, open only one bottle at a time. Store additional bottles without opening until needed for dosing.
At the time of dosing, remove one tablet from the bottle. Immediately close the bottle, secure the cap, and keep the bottle tightly closed.
Keep the desiccant in the bottle.
However, as noted by Managing Editor of Pharmacy Times Laura Silvius, the updated guidelines do not actually say whether or not NDMA has been removed from the product, just that “manufacturing improvements” would address the impurities.7,8

Ranitidine does not appear to be back on the shelves in most retail pharmacies yet, and GoodRx still displays a message saying the 75 mg and 150 mg formulations have been discontinued.9 However, it is available once again as a prescription. Time will tell if retail pharmacies put it back up for purchase as an OTC product.
If you or someone you know experiences heartburn, GERD, or ulcers, consult your provider before trying ranitidine.
References
1 History of Zantac (Ranitidine). Arnold & Itkin Trial Lawyers. Personal Injury Attorneys. 2020.
6 Zantac 360. Opella Healthcare Group SAS.
8 Silvius L. FDA Approves Reformulated Ranitidine After 5-Year Absence. Pharmacy Times. 2025 Nov 25.
*Information presented on RxTeach does not represent the opinion of any specific company, organization, or team other than the authors themselves. No patient-provider relationship is created.

